Electronic Lab Note Book

Electronic LabNote(eLN) software designed for pharmaceutical, biotech, CRO, academic, and industrial laboratories.
Our secure, cloud-based ELN improves research productivity, ensures regulatory compliance, and enables AI-ready data for faster drug discovery and development.
eLabNote helps scientists focus on science instead of documentation, while organizations benefit from secure data management, audit readiness, and seamless collaboration.
What is an Electronic Lab Note (eLN)?
An Electronic Lab Note (eLN) is a digital platform that replaces traditional paper lab notebooks by allowing researchers to record, store, search, and share experimental data securely.
Unlike paper notebooks, eLN enables:
- Structured and unstructured data capture
- Secure, tamper-proof electronic records
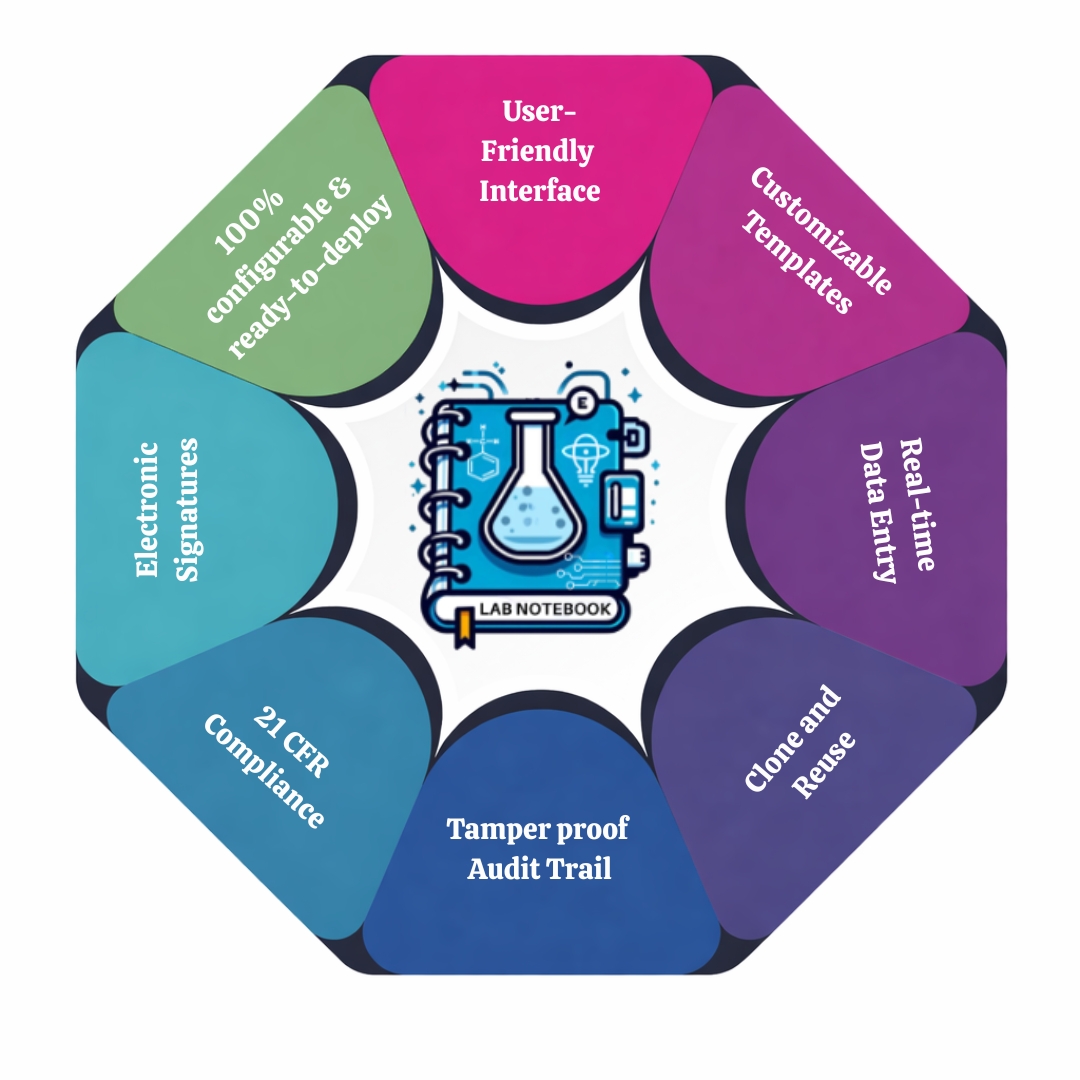
- Full audit trails and electronic signatures
- Easy collaboration across labs and locations
eLabNote offers 90% pre-built, 100% configurable, ready-to-deploy lab notebooks that can be configurable, and can be fully customized to meet the specific needs of any pharmaceutical organization.
Core Features of eLabNote :
Smart Experiment Management
- Clone experiments to reduce data entry time
- Automatic date and time stamping
- Link experiments to raw data files and results
- Lock experiments for audit and IP protection
Advanced Data Capture
- Drag-and-drop files (PDF, images, spectra, reports)
- Manage structured and unstructured lab data
Built-In Analytics & Reporting
- Automated report generation
- Project tracking
Regulatory Compliance & Data Integrity
eLN is built to meet global regulatory requirements for pharmaceutical and life sciences research.
Compliance Standards Supported
- FDA 21 CFR Part 11 compliant ELN
- GxP (GLP, GMP)
- ISO standards
Compliance Capabilities
- Secure closed system with role-based access
- Tamper-proof audit trails with timestamps
- Electronic signatures with non-repudiation
- PDF export
FAIR Data & Digital Transformation
Our ELN fully supports FAIR data principles:
- Findable – Advanced search & metadata
- Accessible – Secure cloud/server access
- Interoperable – APIs and system integrations
- Reusable – Standardized, high-quality data
This ensures your lab data is future-proof, AI-ready, and reusable across projects.
Operational & Business Benefits :
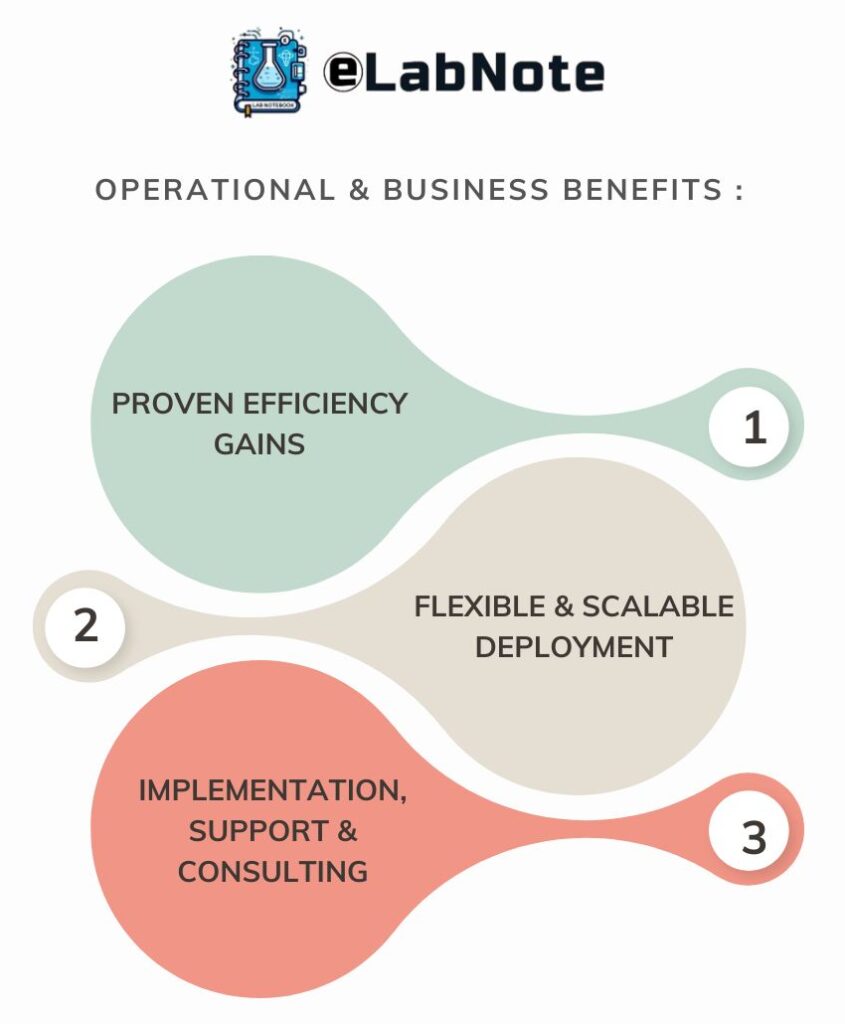
Proven Efficiency Gains
- Reduce lab data entry time by up to 60%
- Faster data retrieval and reporting
- Lower compliance and audit preparation costs
- Improved collaboration and productivity
Flexible & Scalable Deployment
- Cloud-based or on-premise ELN
- No local software installation required
- Low hardware and IT maintenance
- Flexible pricing models
Implementation, Support & Consulting
PharmaDEM provides end-to-end eLN implementation, validation support, and consulting services.
- Rapid onboarding and training
- Lab-specific workflow configuration
- IP, security, and access control setup
- Ongoing customer support
Transform Your Lab with eLabNote
Digitize your lab workflows, ensure regulatory compliance, and unlock the power of data-driven R&D.
